Protein Structure and Function
Introduction:
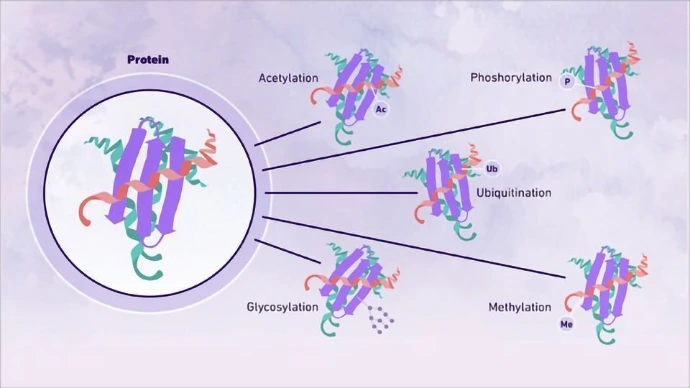
Proteins are essential biological macromolecules responsible for catalysis, signaling, transport, and structural integrity. Their three-dimensional structure dictates function, from enzymatic activity to molecular recognition.
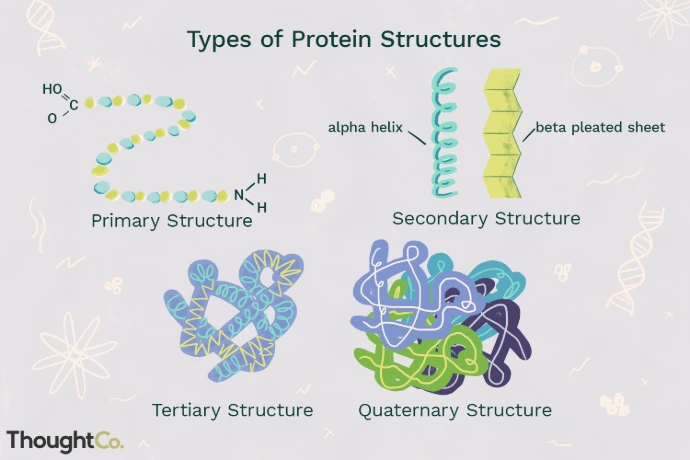
Primary, secondary, tertiary, quaternary structures: The primary structure is the linear sequence of amino acids, determined by the gene sequence. Secondary structures include alpha helices and beta sheets stabilized by hydrogen bonds. Tertiary structure refers to the overall 3D fold, influenced by hydrophobic interactions, disulfide bonds, and electrostatic forces. Quaternary structure involves multiple polypeptide chains assembling into functional units, like hemoglobin.
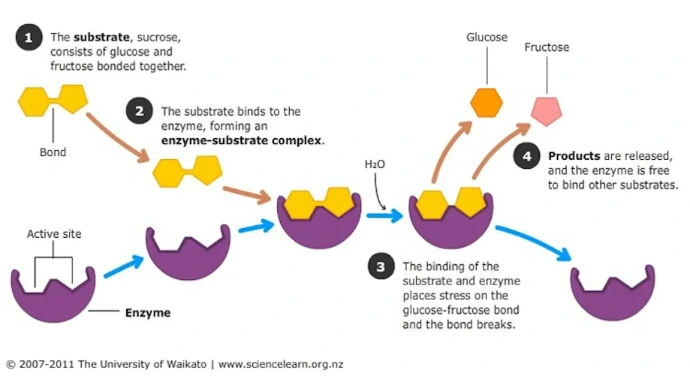
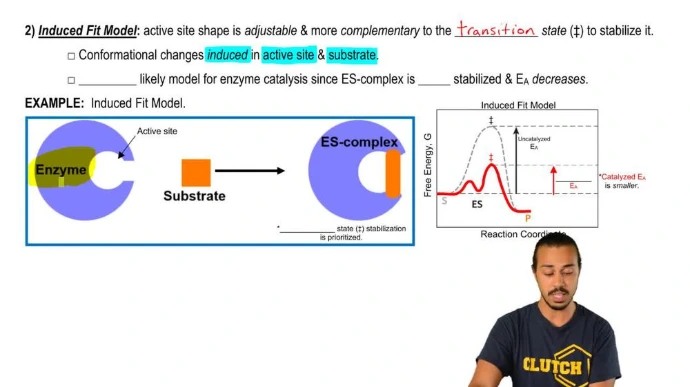
Active sites and functional domains: Active sites are specific regions where substrates bind and reactions occur, often involving key residues like in enzymes. Functional domains are modular units within proteins that perform distinct roles, such as DNA-binding or catalytic domains, allowing for evolutionary reuse.
Conclusion
Understanding protein structure enables medcine design, therapeutic targeting, and synthetic biology innovations.